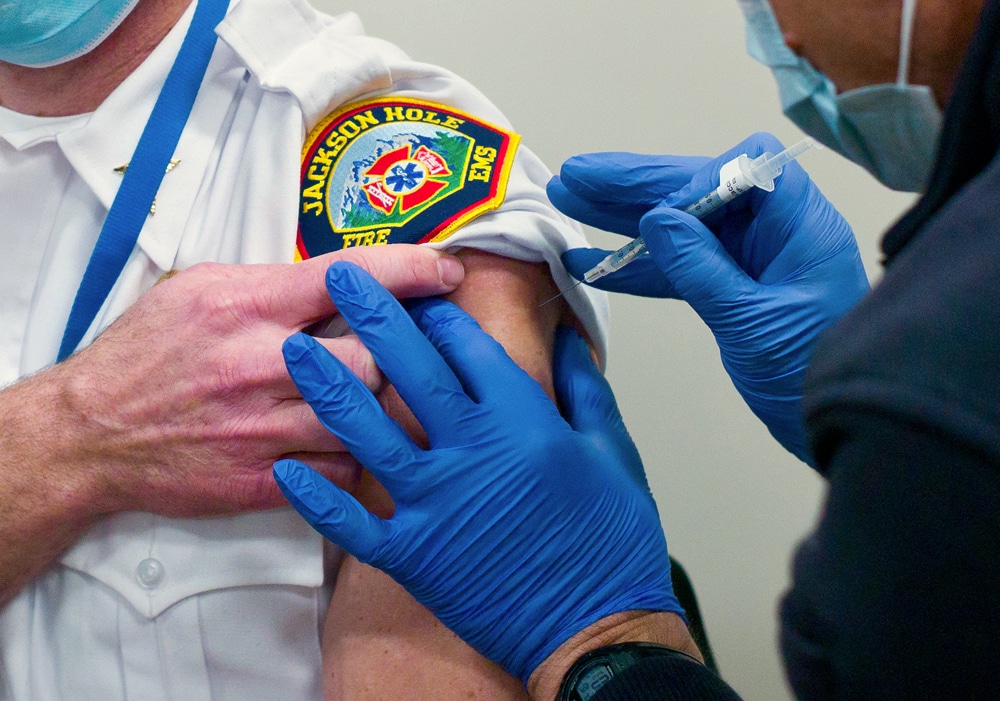
When a nurse pierced emergency room worker Patrick Almond’s right arm with a syringe holding 0.3 milliliters of the Pfizer-BioNTech COVID-19 vaccine in Jackson on Wednesday, it marked the end of a long, cold trip for the concoction named BNT162b2.
Pfizer makes the vaccine at plants in Missouri, Massachusetts, Michigan and Wisconsin and this week shipped doses directly to five locations in Wyoming. Packages containing 975 doses each went to public health departments in Casper and Cheyenne and to hospitals in Cody, Gillette and Jackson.

For all the hoopla surrounding authorization, and now distribution, of the vaccine, its arrival in Wyoming was relatively mundane. There were no escorts into towns by wailing fire trucks or banner-clad pickups, no showy celebration.
“It was just delivered by a FedEx delivery driver,” said Hailey Bloom, spokeswoman with the Casper-Natrona County Health Department.
There was one special instruction, she said. Delivery workers “were told to be careful with this box.”
“This box” was different from other deliveries. Measuring about 2 feet on each side, it held not only 975 potentially life-saving doses of the vaccine but layers of insulation and dry ice. Pfizer designed the containers to keep the vaccine at the required low temperature — between 76 and 112 degrees below zero — for up to 10 days.
“There’s quite a bit of dry ice involved and a tiny bit of vaccine,” Bloom said. BNT162b2’s 975 frozen doses emerge from the packaging in a carton “the size of a small pizza box,” said Kim Deti, spokeswoman for the Wyoming Department of Health.
Between logistics, packaging and special handling, thousands of people contributed to creating and distributing a few milliliters of frozen liquid across the state. After all, BNT162b2 is, in the words of St. John’s Health CEO Paul Beaupre, “liquid gold.”
Tracking all the way
In addition to designing its transport packaging and the specialized temperature-maintenance protocols that go with it, Pfizer tracks each package. “We will have a Pfizer 24/7 control tower monitoring and tracking all shipments,” Pfizer Chairman and CEO Dr. Albert Bourla wrote in a statement announcing the rollout.
The attention to detail is immediately apparent, Natrona’s Bloom said. “The very first thing you see is that log tag telling you this vaccine was maintained or not maintained at the proper temperature,” she said.
The inside of the transport box was “pretty impressive,” said Ken Jarman, director of pharmacy at St. John’s Health. The tracking tag he saw inside was green, “which is good,” he said, and indicated the 195 vials had not experienced a “temperature excursion” that would disqualify them from use.
He pushed a button on the tag to indicate he had received the vaccine. About 15 minutes later he received a confirmation email from Pfizer.
“It was a green light for use.” Jarman said.
A full tray of the vaccine can’t be out of the transport box or its designated super-freezer for more than five minutes. Partial trays can’t be out of the deep freeze for more than three minutes, Bloom said.
But you can’t inject somebody with a substance that’s colder than the average winter temperature at the South Pole. Pharmacists must first thaw BNT162b2, which takes a couple of hours, then dilute it for injection.
Once out of the deep freeze, the vaccine is good for five days, maximum. Health workers prepare it for injection by inverting the vials, each of which contains enough concentrated vaccine for at least five doses, 10 times.
Health workers next draw 1.8 mL of 0.9% saline solution into a transfer syringe and inject that into the vaccine vial. They next use the transfer syringe to withdraw 1.8 mL of air from the vial to equalize pressure, then write down the date and time of dilution.
Dilution triggers another deadline. The reconstituted vaccine must be injected within six hours, Jarman said. Because of that, medical centers have lined up vaccine recipients — for now frontline health workers — in advance.
First jabs
At 2 p.m. Wednesday, Will Smith, paramedic and emergency room doctor in Jackson, rolled up his white EMS uniform sleeve and got jabbed with the first of two doses. “I was not expecting it to happen this week,” he said after receiving his CDC vaccination card. He’ll be back for round two in about three weeks.
Relishing the science that made the “amazing” vaccine, Smith cautioned that BNT162b2 is, seemingly like everything else associated with slowing COVID-19, only one tool.

“The vaccine is going to be one part of the process,” he said. “We know at some point it will decrease our risk.”
That’s an important part of this phase of the pandemic, DOH spokeswoman Deti said. “This gives us some hope there’s a path forward and that feels very good,” she said.
However, it will take a while — months — to get most Wyoming residents vaccinated she said.
“It’s going to take some patience,” she said. “For a while longer we do need people to follow our primary recommendations,” she said, including wearing masks, staying apart and washing hands.
Right now, vaccinated people should still wear masks the same way the unvaccinated do, the federal Centers for Disease Control and Prevention says. That’s because experts aren’t certain whether the vaccine, which guards against COVID-19 disease and symptoms, also prevents infection by or transmission of the virus itself. The vaccine, a modified partial strain of COVID-19 RNA injected into muscle tissue, provokes an immune response that jump-starts antibody production to fight against infection.
Smith and his ER colleague Almond benefited from the first round of vaccines — Wyoming got a total of 4,875 doses — because Jackson was one of the five Wyoming sites with super freezers certified by the state.
“I read about this vaccine six months ago,” St. John’s Beaupre said. “When Pfizer looked like it was getting ahead, that’s when we decided to get the freezer.”
Natrona County also saw the looming need for the special equipment. “We were able to get one early on,” Bloom said. Around the first part of November the county made a quick decision and placed its order.
Later that day, the supplier had sold out, she said.
Because of the specialized transport and storage requirements of the Pfizer vaccine, the first allocations will be administered in the counties where they were distributed, Deti said.
“We made a [distribution] decision based on who could receive and store it,” she said. “It was decided not to move them between counties.”
That exclusivity should pass with the expected emergency-use approval of another vaccine, this one developed by Moderna. It doesn’t require the super-cold storage necessary for the Pfizer product.
With that approval expected this week, “we should have some vaccine in every county next week,” Deti said.
Relief
Wyoming’s DOH has posted information about the vaccine on its website, including the priority list for who’s first in line. The arrival of the vaccine has changed attitudes for perhaps the first time in nine months.
“I felt pretty relieved and excited,” Deti said. “This gives us some hope there’s a path forward and that feels very good.”
DOH’s busy schedule is now infused with a new vigor.
“This is busy in a different way,” she said. “We can take the view that we’re doing something more positive.”
In Natrona County, vaccine logistics are “a welcome challenge” and a move out from the dark, Bloom said. “To be honest, some days it seemed like it wouldn’t come.”
Support independent reporting — donate to WyoFile today
Excited ER worker Almond said he’s also a bit nervous because emergency-use authorization hasn’t answered questions about the long-term effects of inoculation. Risks from COVID-19, however, outweigh that worry, especially considering his pregnant wife at home, he said.
For Beaupre, announcing the pending arrival of the vaccine to hospital workers “was the best Christmas present we could give them.”
A bit of anxiety and stress permeated the pharmacy department at St. John’s when the vaccine arrived after only a 48-hour prep period, Jarman said. But, “it was an exciting and good stress,” he said.
“We are giddy,” Jarman said. “We feel blessed.”

Dear Mr. Thuermer,
Some beautiful.writing here. I’ve been thinking a lot about the logistics of delivering the Pfizer product to rural America the last few days. Posted this on my facebook page. Keep writing.